The Ideal Gas Law (Clapeyron-Mendeleev equation)
Gas Properties by PhET Interactive Simulations, University of Colorado Boulder, licensed under CC-BY-4.0 (https://phet.colorado.edu)
Project name: The Ideal Gas Law (Clapeyron-Mendeleev equation)
This virtual laboratory is intended for use in chemistry classes on the following topics:
- 10th grade. Chapter VI. “Kinetics”
Goals:
- to explore the Ideal Gas law using the PHET Interactive Simulation: Gas Properties.
- to integrate the relationships from Boyle’s, Charles’s, and Gay-Lussac’s laws to understand the Ideal Gas Law (PV = nRT).
Theory
Gas laws describe the behavior of gases in different conditions. Understanding these laws helps us predict how gases will respond when we change their pressure, volume, or temperature.
Ideal Gas Law
The Ideal Gas Law, also known as the Clapeyron-Mendeleev Equation combines several simpler gas laws into one equation:
PV = nRT
This equation shows how pressure (P), volume (V), number of particles (n), the gas constant (R), and temperature (T) are related.
- P (pressure) is the force that gas particles exert on the walls of their container.
- V (volume) is the space that the gas occupies.
- n (number of moles) is the amount of gas.
- R (gas constant) is a fixed value that makes the units work out correctly (8.31 J/(mol·K)).
- T (temperature) is a measure of how fast the gas particles are moving.
Boyle’s Law (P ∝ 1/V at constant T)
Boyle’s Law states that if the temperature of a gas is kept constant, its pressure and volume are inversely proportional. This means:
- When the volume decreases, the pressure increases.
- When the volume increases, the pressure decreases.
Equation: P₁V₁ = P₂V₂
Charles’s Law (V ∝ T at constant P)
Charles’s Law states that if the pressure of a gas is kept constant, its volume and temperature are directly proportional. This means:
- When the temperature increases, the volume increases.
- When the temperature decreases, the volume decreases.
Equation: V₁/T₁ = V₂/T₂
Gay-Lussac’s Law (P ∝ T at constant V)
Gay-Lussac’s Law states that if the volume of a gas is kept constant, its pressure and temperature are directly proportional. This means:
- When the temperature increases, the pressure increases.
- When the temperature decreases, the pressure decreases.
Equation: P₁/T₁ = P₂/T₂
Practical part

- Open the Simulation. This tool models the behavior of gas particles in a container, helping visualize concepts like pressure, volume, and temperature.
2. Get familiar with the components of the simulation: the particle system (gas molecules), the container, and the adjustable parameters such as pressure (P), volume (V), and temperature (T).
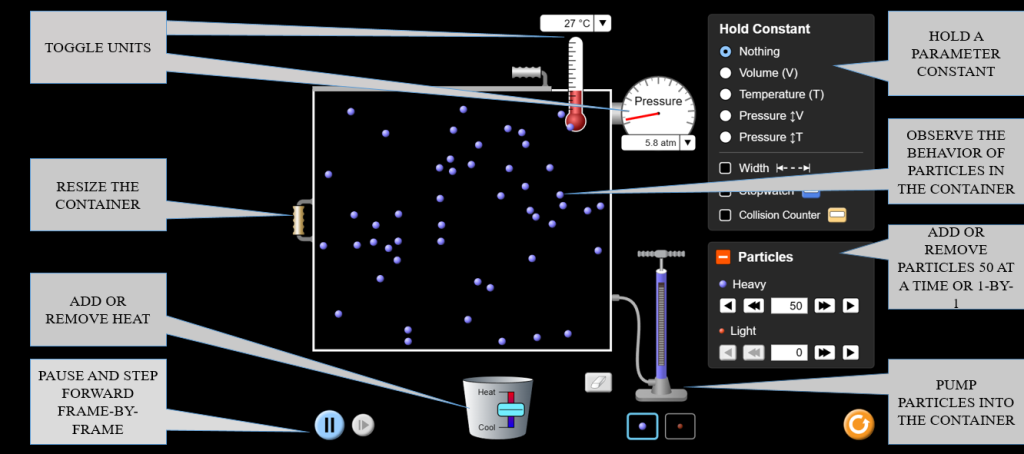
Exploring Boyle’s Law (P ∝ 1/V at constant T)
- Navigate to the Ideal screen within the simulation. Set Initial Conditions: Start with a fixed number of particles (N) and a constant temperature (T). You will observe how pressure changes with volume.
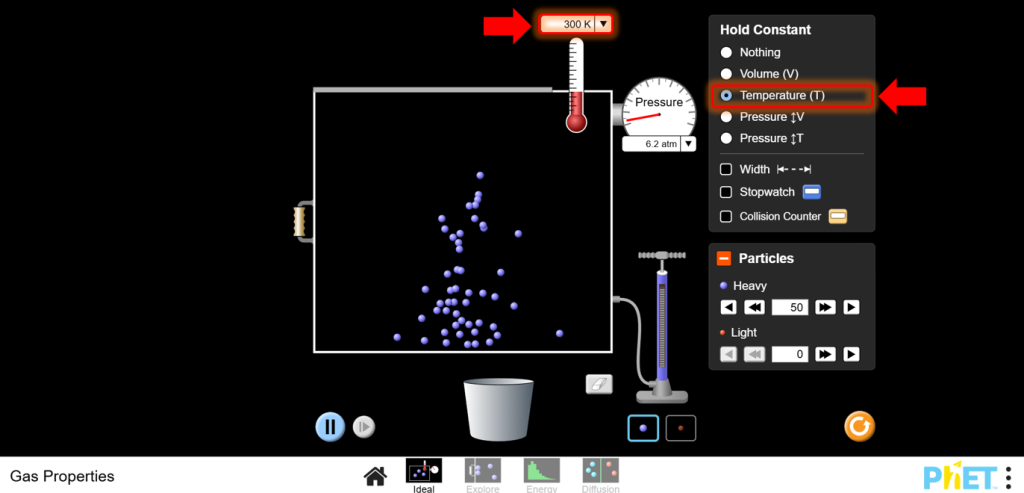
4. Adjust Volume:
a.Use the left wall of the container to change the volume. Reduce the volume and observe the pressure increase.
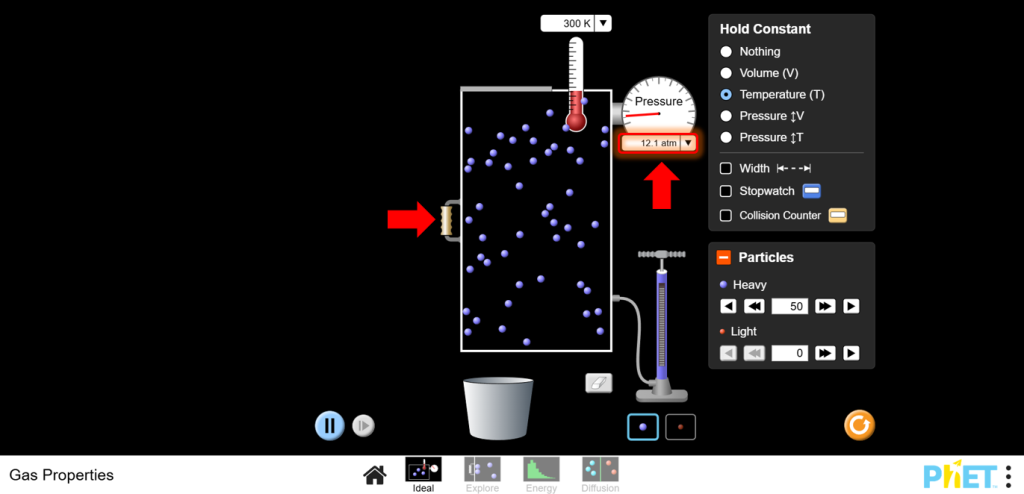
b.Increase the volume and observe the pressure decrease.
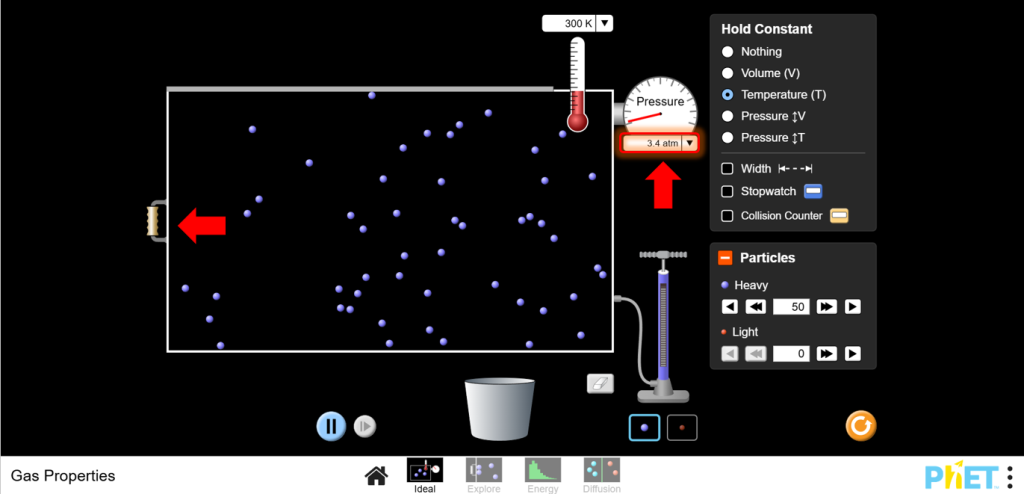
5. Discussion: Boyle’s Law states that pressure and volume are inversely proportional when temperature is constant. Record your observations and plot P vs. 1/V to see the inverse relationship.
Exploring Charles’s Law (V ∝ T at constant P)
- Set Up for Charles’s Law:Move to the Ideal screen. Keep the pressure constant by adjusting the left wall of the container as needed.
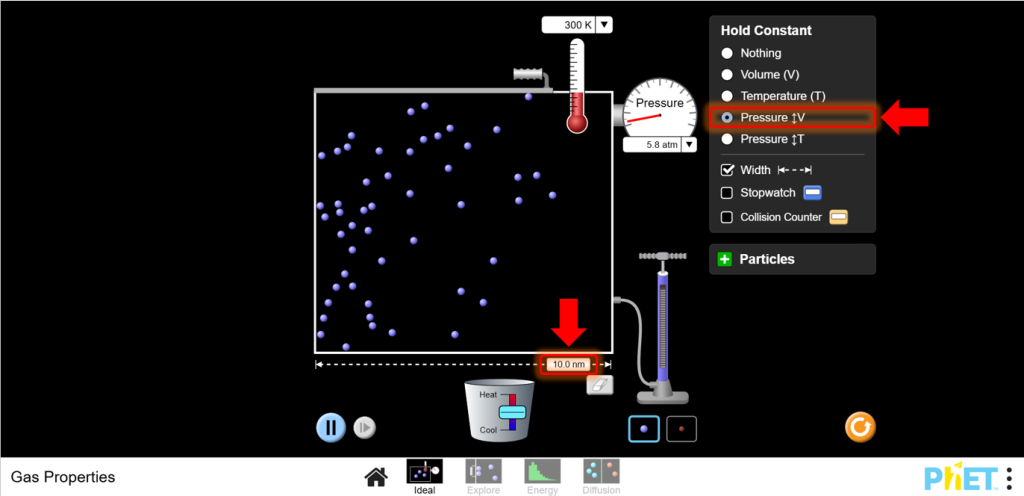
7. Vary Temperature:
a. Increase the temperature using the heater.Note how the volume of the container changes with temperature. Observe that as temperature increases, volume increases
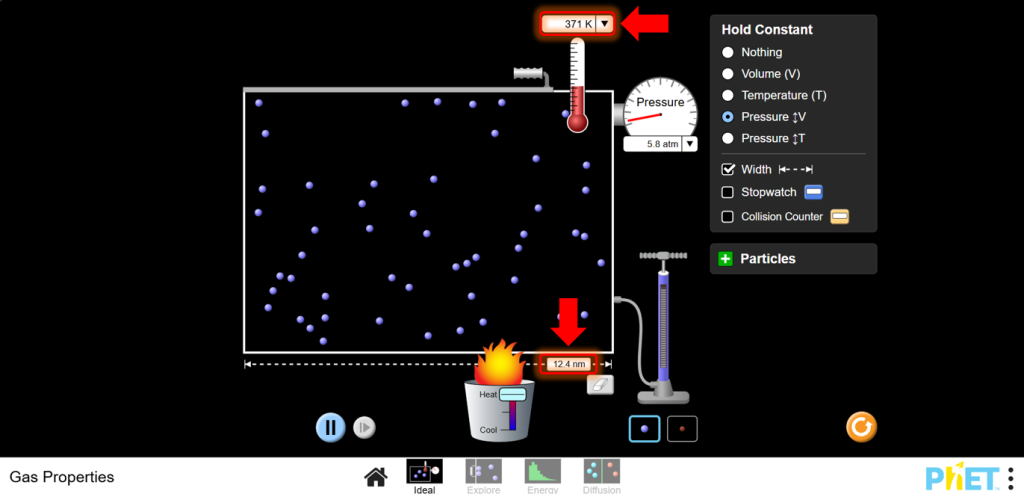
b. Decrease the temperature using the cooler. Observe that as temperature decreases, volume decreases too.
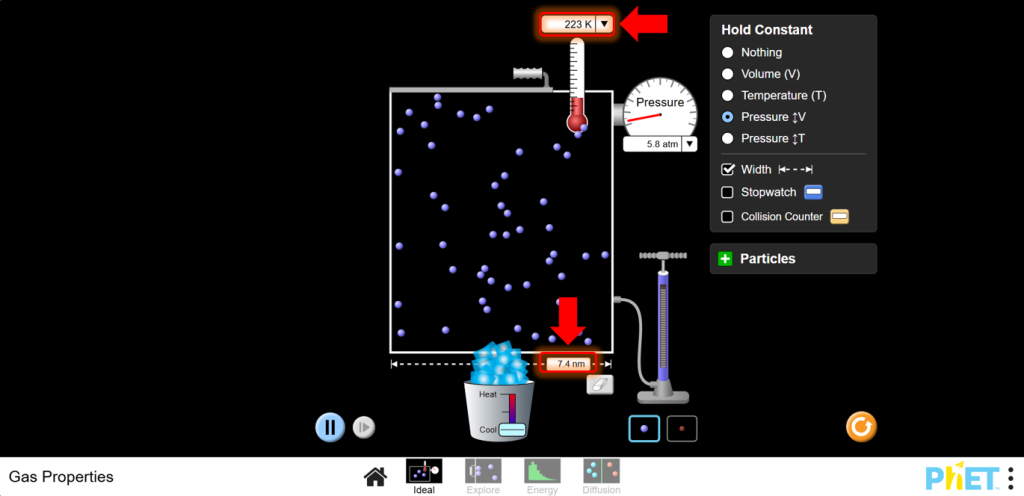
8.Discussion: Charles’s Law shows a direct relationship between temperature and volume when pressure is constant. Plot V vs. T and observe the linear relationship.
Exploring Gay-Lussac’s Law (P ∝ T at constant V)
- Set Up for Gay-Lussac’s Law:Stay on the Ideal screen. Keep the volume constant by not adjusting the left wall of the container.
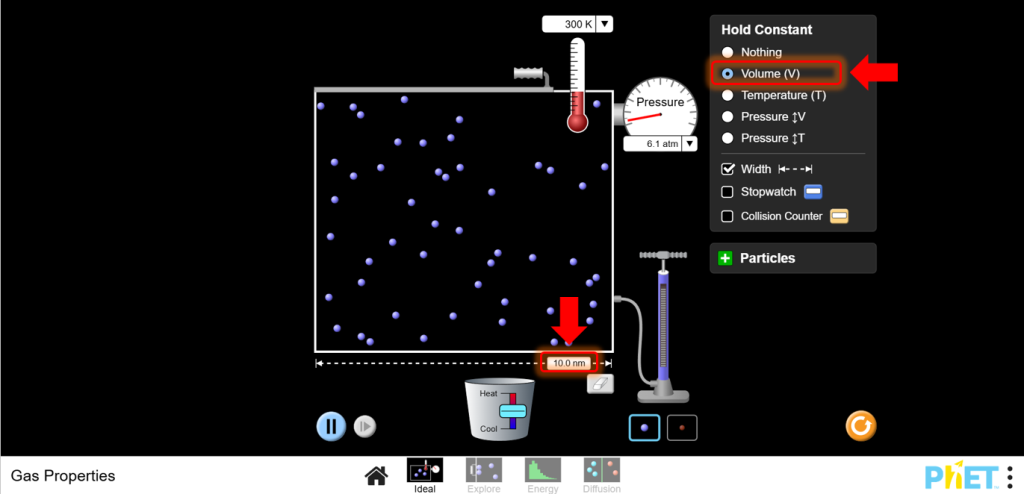
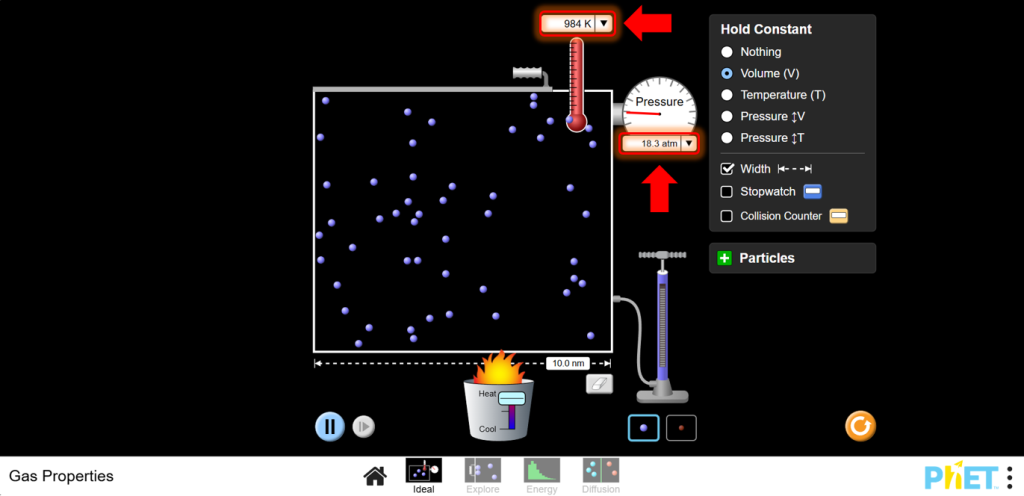
10. Vary Temperature:
a. Use the heater to increase the temperature. Monitor how the pressure changes as the temperature changes. You should see that as temperature increases, pressure increases.
b.Use the cooler to decrease the temperature. You should see that as temperature decreases, pressure decreases too.
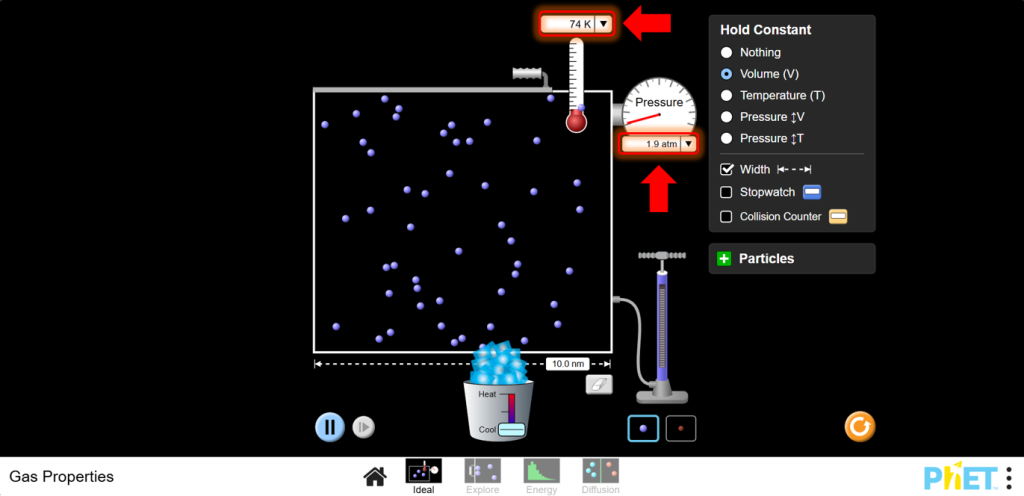
11. Discussion: Gay-Lussac’s Law states a direct relationship between pressure and temperature when volume is constant. Plot P vs. T to visualize this direct proportionality.
Applying the Ideal Gas Law (PV = nRT)
- Summarize Relationships: Explore how Boyle’s, Charles’s, and Gay-Lussac’s laws combine into the Ideal Gas Law.
- Conduct an Experiment:
a. Choose a fixed number of particles.
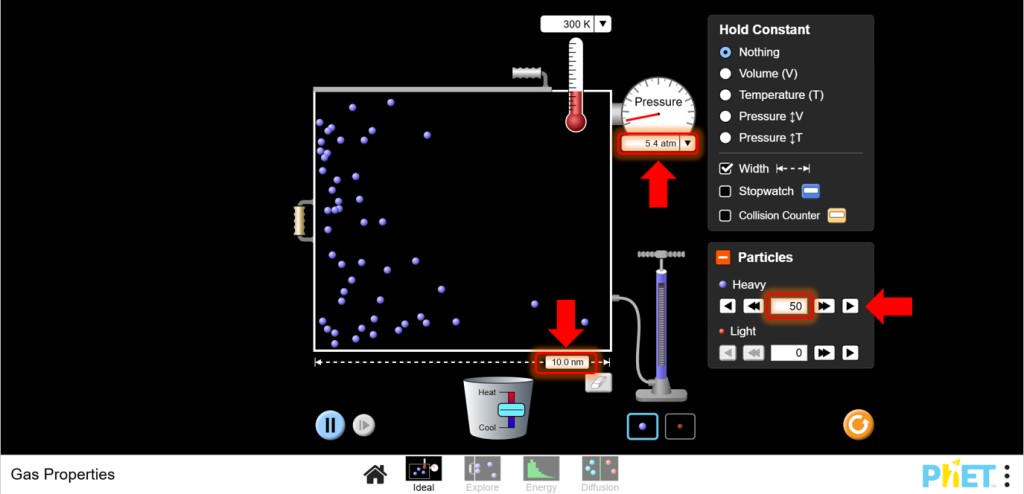
b. Adjust one variable (e.g., volume) and observe changes in pressure and temperature. Predict changes based on the Ideal Gas Law before making adjustments.
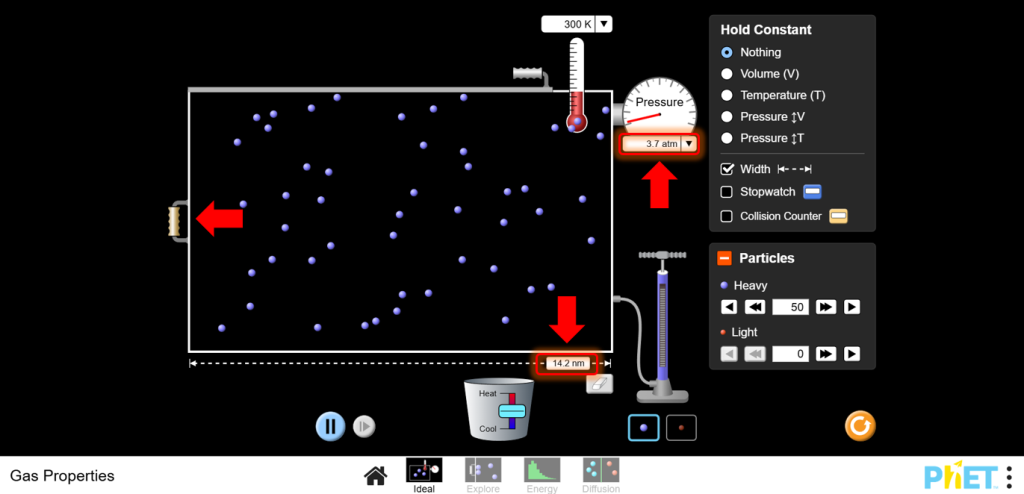
14. Calculation Practice:You can work with different scenarios to calculate unknown quantities using PV = nRT, reinforcing the mathematical relationship.
Conclusion
Using the PhET “Gas Properties” simulation provided a hands-on approach to learning the Ideal Gas Law and its component relationships: Boyle’s Law, Charles’s Law, and Gay-Lussac’s Law. By engaging in interactive experiments, students were able to visualize and understand the behavior of gases under different conditions. The simulation effectively demonstrated the principles of these gas laws, helping students to connect theoretical concepts with practical observations.